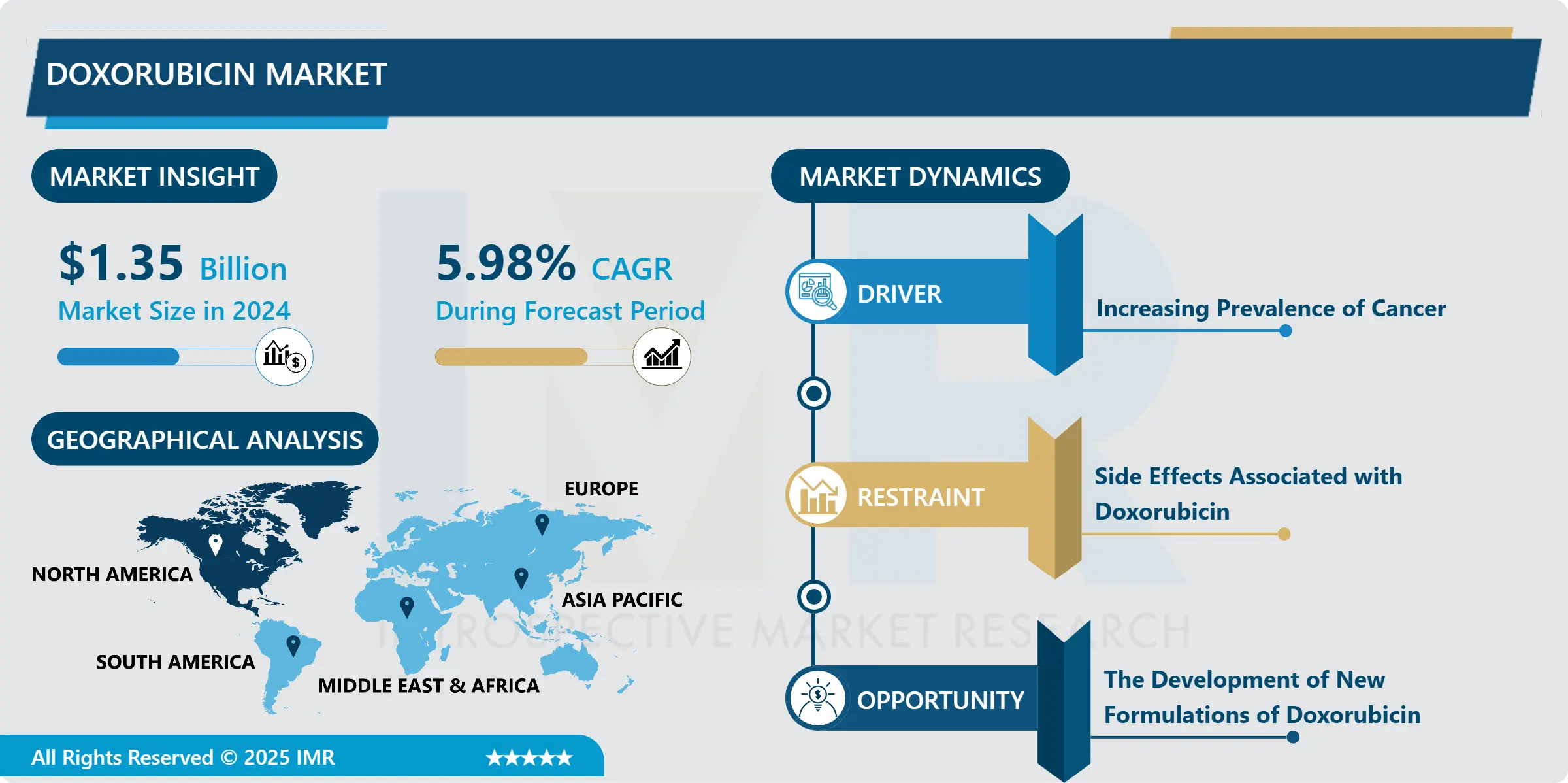
The global doxorubicin market, valued at USD 1.35 billion in 2024, is projected to expand steadily and reach USD 2.56 billion by 2035, driven by a CAGR of 5.98% between 2025 and 2035. This growth is fueled by a rising global cancer burden, broader adoption of chemotherapy regimens, and increasing development of safer, next-generation doxorubicin formulations designed to reduce toxicity.
Quick Market Highlights
2023 Market Size: ~ US$ 1.27 Bn
2032 Projection: ~ US$ 2.13 Bn
Forecast CAGR (2024–2032): ~ 5.90%
Key Formulations: Lyophilized Powder, Doxorubicin Injection
Major Applications: Breast Cancer, Leukemia, Lymphoma, Bladder Cancer, Kaposi Sarcoma, Others
Distribution Channels: Hospital Pharmacies, Retail Pharmacies, E-Commerce
Leading Companies: Pfizer Inc., Cipla, Zydus Group, Sun Pharma, Johnson & Johnson
Top Regional Market: North America, supported by high cancer incidence and comprehensive oncology infrastructure
Primary Restraint: Cardiotoxicity and related side-effect profile
Major Opportunity: Liposomal and nanoparticle-based formulations to reduce toxicity
Key Market Questions: Trends & Opportunities
• Can innovative formulations improve doxorubicin’s safety profile?
Yes. Liposomal and nanoparticle-based delivery systems present a major opportunity by reducing systemic toxicity—particularly cardiotoxicity—allowing for safer use in long-term or high-dose regimens.
• Is generic expansion improving access?
With major pharmaceutical manufacturers ramping up production, generic doxorubicin is becoming more accessible, especially in emerging markets. This widens patient reach but introduces pricing pressure.
• Will combination therapies drive future demand?
Absolutely. Doxorubicin remains a core component of multi-agent protocols such as R-CHOP, R-CHP, and other lymphoma and leukemia regimens. Ongoing trials, including Genentech’s polatuzumab combinations, underscore its continued relevance.
Expert Insight
“Doxorubicin remains a cornerstone in oncology due to its broad-spectrum efficacy. But its future growth hinges on innovation—particularly formulations that mitigate cardiac risk. Companies that deliver safer versions will gain competitive advantage and enhance patient outcomes.”
— Dr. Mira Saxena, Principal Consultant, Introspective Market Research
Market Segmentation
1. By Formulation: Lyophilized Powder & Doxorubicin Injection
Doxorubicin Injection currently dominates the market due to its widespread use in hospital-based chemotherapy and its suitability for multi-agent treatment regimens. It offers dosing flexibility, rapid administration, and high adoption in advanced cancer centers.
Lyophilized Powder, however, is gaining traction for its stability and longer shelf life—key advantages for markets with limited cold-chain infrastructure. It supports precise reconstitution and customized dosing, making it highly relevant for expanding oncology services in developing regions.
Together, these formulations provide complementary benefits and ensure robust global availability.
2. By Application: Extensive Oncology Adoption
According to Introspective Market Research, doxorubicin is utilized across a broad spectrum of cancers:
Breast Cancer: Largest revenue contributor; widely used in AC (Adriamycin + Cyclophosphamide) regimens.
Leukemia & Lymphoma: Integral to standard treatments such as R-CHOP and ABVD, ensuring strong and recurring demand.
Kaposi Sarcoma: Important in regions with high HIV prevalence.
Other Cancers: Includes sarcomas, ovarian cancer, and pediatric tumors, with anticipated growth as clinical trials explore safer formulations.
Its versatility across tumor types solidifies doxorubicin’s role as a backbone of contemporary cancer therapy.
3. By Distribution Channel: Hospital Dominance
Hospital Pharmacies: Largest segment; driven by the need for specialized handling and administration in controlled oncology settings.
Retail Pharmacies: Slowly expanding with outpatient chemotherapy and decentralized cancer care models.
E-Commerce: Emerging in select markets, particularly for procurement of oncology drugs by certified facilities.
Market Challenges
Cardiotoxicity: Limits cumulative dosage; requires frequent monitoring and expert management.
Regulatory Complexity: Advanced formulations face stringent multi-phase validation.
Pricing Pressure: Generics drive competition and may compress margins.
Handling Costs: Specialized storage and preparation elevate operational expenses.
Case Study: Advancing Safety Through Liposomal Doxorubicin — PharmaNova’s Breakthrough
Background
PharmaNova, an innovation-focused oncology company, recognized the unmet need for a safer doxorubicin formulation due to high cardiotoxicity risks. Older patients and those with comorbidities often struggled to complete treatment, limiting clinical outcomes.
Determined to address this challenge, PharmaNova embarked on developing a PEGylated liposomal formulation designed to deliver powerful anti-tumor activity with reduced cardiac strain.
The Challenge
Conventional doxorubicin is effective but restricted by:
Cumulative cardiac toxicity
Higher risks among elderly patients or those with hypertension, diabetes, or pre-existing heart disease
Increased costs linked to cardiac monitoring and cardio-oncology consultations
Treatment interruptions due to adverse events
PharmaNova aimed to create a formulation that improved safety while proving cost-effective for providers and payers.
The Solution: PEGylated Liposomal Doxorubicin
The company invested in nanotechnology to develop a formulation that:
Reduced cardiac exposure
Prolonged circulation time
Enhanced tumor targeting through the EPR effect
Minimized systemic toxicity
Clinical Findings
Trials involving breast cancer patients with cardiac risk factors showed:
Significant reductions in LV dysfunction and cardiac biomarkers
Tumor response rates comparable to conventional doxorubicin
Higher patient compliance and fewer treatment interruptions
These results paved the way for regulatory approval in major markets.
Outcome & Market Impact
1. Better Patient Outcomes & Treatment Continuity
The new formulation resulted in:
Fewer cardiac adverse events
Higher treatment completion rates
Reduced emergency cardiac interventions
Improved quality of life
2. Rapid Uptake in Oncology Centers
North America and Europe adopted the product quickly due to stringent focus on cardio-oncology safety and high demand for safer chemotherapy options.
3. Strong Commercial Success
Even with premium pricing, hospitals found overall cost savings due to:
Lower monitoring burdens
Fewer hospitalizations
Enhanced adherence