Troponin Market To Get Great Push By 2032, Driven by High-Sensitivity Assays, Point-of-Care Expansion, and Rising Cardiovascular Disease Burden.
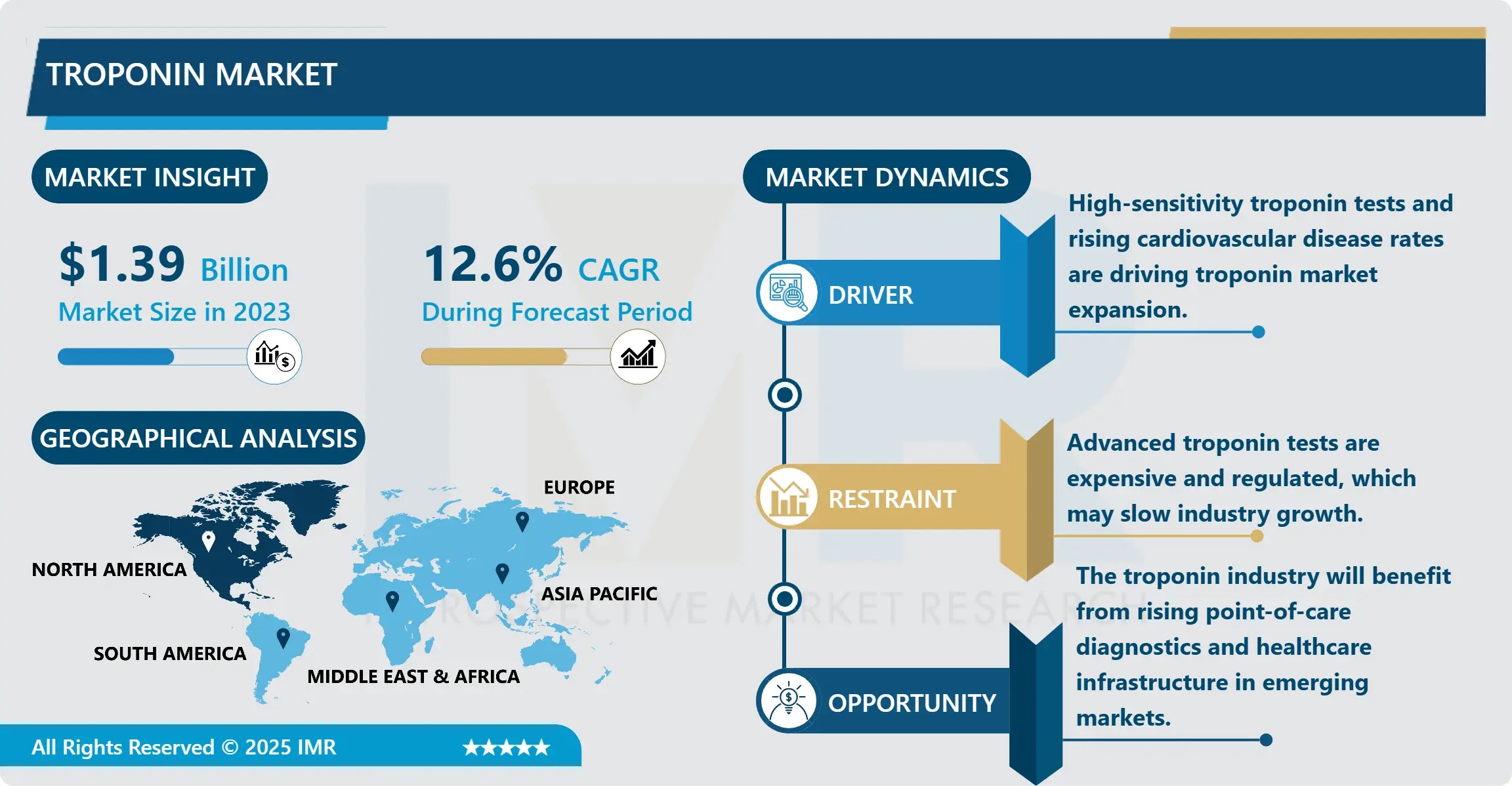
The Global troponin market is projected to surge from USD 1.39 billion in 2023 to USD 4.04 billion by 2032, registering a robust CAGR of 12.6% over the forecast period (2024–2032), according to a new in-depth report by Introspective Market Research. This accelerated growth reflects a paradigm shift in cardiac diagnostics—where troponin testing has evolved from a confirmatory tool for myocardial infarction to a cornerstone of precision cardiovascular risk assessment, early intervention, and real-time clinical decision-making. Key catalysts include the global rise in cardiovascular diseases (CVDs), rapid adoption of high-sensitivity cardiac troponin (hs-cTn) assays, integration of troponin into chest pain protocols, and the widespread rollout of portable point-of-care (POC) platforms in ambulances, rural clinics, and home-care settings.
With CVDs remaining the world’s leading cause of death—responsible for an estimated 17.9 million fatalities annually—the demand for faster, more accurate, and accessible cardiac biomarker tools has never been greater. Troponin I and Troponin T, once used primarily in hospital labs, now anchor triage algorithms in emergency departments worldwide and are increasingly deployed for longitudinal monitoring in heart failure and post-chemotherapy cardiotoxicity management.
Quick Insights: Troponin Market Snapshot (2023–2032)
- 2023 Market Value: USD 1.39 Billion
- 2032 Projected Value: USD 4.04 Billion
- Forecast CAGR (2024–2032): 12.6%
- Dominant Product Segment: Troponin T (due to superior long-term prognostic utility and standardization in chronic cardiac profiling)
- Largest Application Area: Myocardial Infarction (MI)
- Top End User Segment: Hospitals (driven by ER/ICU demand)
- Leading Region: North America
- High-Growth Trend: Point-of-Care Testing (POCT), home monitoring, AI-integrated interpretation
- Key Players: Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Beckman Coulter, bioMérieux SA, Thermo Fisher Scientific, Radiometer Medical, Mitsubishi Chemical Europe
How Is Troponin Testing Evolving Beyond the Lab into the Frontlines of Cardiac Care?
What innovations are turning a blood test into a real-time lifeline across the care continuum?
The answer lies in three converging revolutions:
- High-Sensitivity Assay Standardization: Modern hs-cTnI and hs-cTnT platforms can detect concentrations as low as 1–5 ng/L—enabling rule-in/rule-out of acute MI in under one hour (vs. 6–12 hours with conventional assays). ESC and AHA now endorse 0/1-hour and 0/2-hour algorithms, drastically reducing ED overcrowding and misdiagnosis. Roche’s Elecsys® hs-cTnT and Abbott’s ARCHITECT STAT hs-cTnI lead in clinical validation, with >99% negative predictive value (NPV) when used in protocol-driven workflows.
- Democratization via Point-of-Care Platforms: Portable analyzers like Siemens’ Atellica VTL and Radiometer’s AQT90 FLEX bring lab-grade accuracy to ambulances and community clinics—cutting time-to-treatment by up to 45 minutes. Emerging microfluidic POC devices (e.g., Response Biomedical’s RAMP® 2.0) require <50 µL of capillary blood and deliver results in <15 minutes, making them ideal for low-resource and home-monitoring applications.
- Expansion Beyond Acute MI: Troponin assays are now integral to:
- Heart Failure Prognostication: Serial hs-cTnT levels predict 1-year mortality in HFrEF/HFpEF.
- Oncocardiology: Monitoring for anthracycline- or HER2-targeted therapy–induced myocardial injury.
- Chronic Kidney Disease (CKD) Risk Stratification: Elevated troponin in CKD patients correlates with silent ischemia and sudden cardiac death.
“Troponin is no longer just a ‘rule-out MI’ test—it’s becoming the vital sign of myocardial health,” says Dr. Nathan K. Reyes, Principal Consultant, Cardiovascular Diagnostics & Digital Health at Introspective Market Research. “We’re moving toward a future where a single hs-cTnT measurement, contextualized by AI with ECG, clinical history, and renal function, generates a real-time ‘cardiac risk score’—streamlining decision pathways from ER triage to outpatient follow-up. The most exciting frontier is preventive serial monitoring: wearable-integrated microsampling devices could enable weekly troponin tracking in high-risk populations—transforming cardiology from reactive to predictive. This shift demands not just better assays, but seamless EHR integration, clinician education, and equitable access.”
Segmentation Spotlight: Troponin T Leads, Hospitals Anchor, MI Dominates
By Product Type: Troponin T Maintains Edge in Prognostic Utility
While Troponin I offers excellent sensitivity and manufacturer-specific assay flexibility (e.g., Abbott, Siemens), Troponin T—primarily driven by Roche’s standardized global platform—holds the largest market share due to its validated performance in chronic disease monitoring and superior inter-laboratory reproducibility. Troponin Complex assays (detecting both I and T simultaneously) are emerging for complex cases like myocarditis or post-transplant surveillance.
By Application: Myocardial Infarction Remains Core, but Heart Failure Gains Traction
Myocardial Infarction commands the largest application segment (>58% of volume), underpinned by universal guideline adoption (ESC, AHA/ACC). However, Acute Coronary Syndrome (ACS) and Congestive Heart Failure (CHF) are the fastest-growing segments—fueled by data linking even minor troponin elevations to adverse outcomes in stable outpatients. Cardiomyopathy monitoring (e.g., in chemotherapy, sepsis, or viral infections) is a strategic growth zone, particularly in oncology and critical care settings.
By End User: Hospitals Lead, but Home Care Emerges as High-Potential Frontier
Hospitals account for ~72% of market revenue—especially emergency departments, cath labs, and ICUs. Diagnostic laboratories serve the ambulatory and referral ecosystem with centralized hs-cTn testing. Critically, Home Care Settings are gaining momentum: remote troponin monitoring for heart failure patients reduced 30-day readmissions by 22% in a 2024 JAMA Cardiology pilot—spurring CMS interest in reimbursement expansion.
Regional Leadership: North America Dominates, APAC Accelerates
North America leads in market value and innovation adoption. The U.S. alone contributes over 51% of global revenue, propelled by high CVD prevalence, mature insurance coverage (including Medicare for serial testing in CHF), and early uptake of hs-cTn protocols. The FDA’s 2024 guidance on POC troponin device validation further accelerated decentralized testing adoption.
Europe reflects strong standardization momentum—especially in Germany and the UK, where national chest pain pathways mandate hs-cTnT (Roche) as the preferred analyte. EU IVDR compliance has elevated quality benchmarks, though rollout complexity temporarily slowed smaller lab transitions.
Asia Pacific is the highest-growth region (14.3% CAGR), driven by rising CVD burden in China and India, government investments in emergency care infrastructure, and partnerships between global IVD players and local distributors (e.g., Roche with Bhatia Diagnostics, Abbott with Metropolis). Japan and South Korea are pioneering AI-assisted troponin interpretation in telecardiology networks.
Breakthroughs & Strategic Moves by Industry Leaders
- Roche Diagnostics launched Elecsys® hs-cTnT 2.0—a next-gen assay with 3x lower limit of detection (LoD: 1.2 ng/L) and automated delta-check analytics for serial trend interpretation.
- Abbott Laboratories integrated its i-STAT™ Alinity POC system with Epic EHR via FHIR API—enabling real-time troponin results to auto-populate clinical decision support alerts in ED workflows.
- Siemens Healthineers introduced Atellica Solution with AI-Predict Module, combining hs-cTnI results with NT-proBNP and ECG data to generate 48-hour ACS risk stratification scores.
- Radiometer Medical expanded its AQT90 FLEX platform with a Troponin T + NT-proBNP Combo Cartridge—approved for pre-hospital use in European ambulances, reducing false negatives by 18%.
- Response Biomedical received FDA Breakthrough Device designation for its CardioCheck® POC—a smartphone-connected microfluidic chip delivering lab-equivalent hs-cTnI in 12 minutes from fingerstick blood.
Cost Pressures and Pathways to Sustainable Scale
Despite clinical value, cost remains a barrier—particularly for hs-cTn reagents (USD 8–15/test vs. USD 3–6 for conventional). Key efficiency strategies include:
- Protocol-Driven Test Reduction: 0/1-hour algorithms cut repeat testing by 50–70%, saving ~USD 120 per patient encounter.
- POC in Low-Acuity Settings: Deploying POC in urgent care/primary care avoids unnecessary ED transfers—saving ~USD 1,200 per diverted case.
- Bundled Reimbursement Models: CMS’ Cardiac Biomarker Panel Pilot reimburses hs-cTn + NT-proBNP + D-dimer as a single code (CPT 87999), improving lab margins.
- Open-Platform Calibration: Emerging efforts (e.g., IFCC Task Force) aim to harmonize cTnI assays across vendors—reducing hospital switching costs and enabling competitive tendering.
Benefits Extend Beyond Diagnosis: Systemic Impact and Preventive Value
Strategic troponin deployment delivers cascading advantages:
✔ Reduced Mortality: 0/1-hour protocols lower 30-day MI mortality by 19% through faster reperfusion.
✔ Lower Hospitalization Costs: Early rule-out slashes ED length-of-stay by 3.2 hours on average—freeing ~1.4M bed-days annually in the U.S. alone.
✔ Improved Resource Allocation: AI-augmented troponin triage reduces unnecessary coronary angiograms by 27%.
✔ Enhanced Chronic Disease Management: Monthly hs-cTnT monitoring in heart failure clinics improves medication adherence and reduces HF admissions by 2.1/year per patient.
About the Report
“Troponin Market Analysis and Forecast 2024-2032” provides a rigorous, multi-dimensional analysis across product type, application, end user, and region. Highlights include:
- 10-year revenue forecasts (2017–2032) by assay type (hs-cTnI, hs-cTnT, conventional)
- Technology benchmarking: LoD, turnaround time, automation compatibility
- Regulatory pathway mapping (FDA, CE-IVDR, PMDA, CDSCO)
- Competitive profiling of 12+ IVD leaders, including pipeline heatmaps and partnership ecosystems
Download a Free Sample Report or Schedule a Custom Briefing
Hospital administrators, laboratory directors, IVD manufacturers, investors, and policymakers can access the full report—including comparative market sizing tables, POC adoption curves, and regional reimbursement analysis—via the following links:
🔗 Free Sample Report: https://introspectivemarketresearch.com/request/20102
About Introspective Market Research
Introspective Market Research(IMR) is a globally recognized provider of data-driven intelligence across in vitro diagnostics, cardiology, and precision medicine. Our methodology combines primary interviews with 150+ clinical and lab stakeholders, real-world claims data triangulation, and proprietary forecasting models to deliver strategic, evidence-based insights. All reports include dynamic scenario analysis and are updated quarterly to reflect clinical guideline updates and regulatory shifts.
Media Contact
Dr. Elena M. Torres
Senior Director, Global Communications
Introspective Market Research
Email: info@introspectivemarketresearch.com
Phone: +91 91753-37569.
Website: https://introspectivemarketresearch.com