Hepatitis Drugs Market to Reach USD 30.79 Billion by 2032 Amid Global Elimination Push-IMR Report.
Hepatitis Drugs Market to Reach USD 30.79 Billion by 2032 Amid Global Elimination Push-IMR Report.
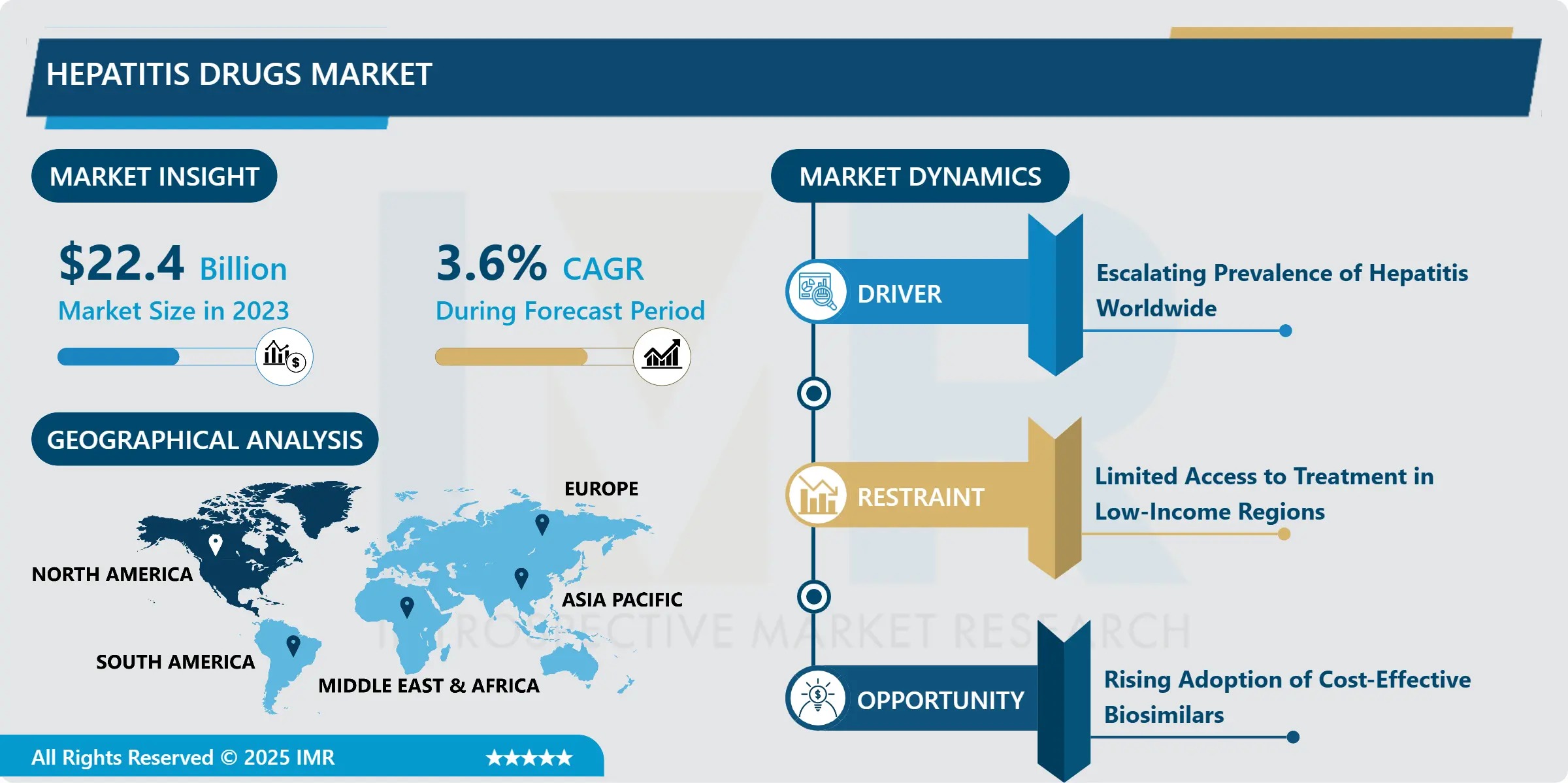
The Global Hepatitis Drugs Market, valued at USD 22.40 Billion in 2023, is projected to grow to USD 30.79 Billion by 2032, advancing at a CAGR of 3.60% from 2024 to 2032, according to a new comprehensive analysis by Introspective Market Research.
While modest compared to hyper-growth biotech segments, this steady expansion reflects a maturing yet high-impact landscape-one increasingly defined by functional cures, access-driven innovation, and strategic geographic rebalancing. With 296 million people living with chronic hepatitis B (HBV) and 58 million with hepatitis C (HCV) globally (WHO, 2024), the clinical and economic imperative remains urgent: untreated viral hepatitis causes ~1.1 million deaths annually-more than HIV and malaria combined.
Growth is now propelled not by volume alone, but by precision: next-generation direct-acting antivirals (DAAs) achieving >99% sustained virologic response (SVR) in HCV; long-acting nucleos(t)ide analogues (NUCs) enabling once-weekly HBV dosing; and rising biosimilar penetration expanding affordability in LMICs. Simultaneously, WHO’s 2030 elimination targets-and national screening campaigns in India, Egypt, and Pakistan-are converting latent prevalence into diagnosed, treatable demand.
Quick Insights: Hepatitis Drugs Market at a Glance
- 2023 Market Size: USD 22.40 Billion
- 2032 Projected Value: USD 30.79 Billion
- CAGR (2024–2032): 3.60%
- Dominant Drug Class: Direct-Acting Antivirals (DAAs)->60% share, driven by pan-genotypic efficacy, oral dosing, and short-duration (8–12 week) regimens
- Largest Disease Segment: Hepatitis B Drugs-accounts for ~52% of revenue (vs. ~38% for HCV), reflecting chronicity, lifelong treatment needs, and higher drug pricing
- Top Regional Market: North America (43.7% share in 2023), led by the U.S.
- Fastest-Growing Region: Asia Pacific-projected CAGR of 4.9%, fueled by mass screening, price-controlled tenders, and rising private insurance coverage
- Key Players: Gilead Sciences, AbbVie Inc., Bristol-Myers Squibb, Johnson & Johnson, Merck & Co., Roche, GlaxoSmithKline, Novartis AG, Dr. Reddy’s Laboratories, Zydus Cadila
If Hepatitis C Is Now Curable, Why Is the Market Still Growing-And Where’s the Next Frontier?
The answer lies in three converging realities:
- Diagnosis Gap → Treatment Gap: Only ~21% of HBV and ~22% of HCV cases are diagnosed globally (WHO, 2024). National screening programs-like India’s Ayushman Bharat Viral Hepatitis Elimination Initiative and Egypt’s 100 Million Seha campaign-are rapidly closing this gap, driving first-wave treatment surges.
- Biosimilars Are Reshaping Access Economics: The patent expiry of landmark DAAs (e.g., sofosbuvir, ledipasvir) has catalyzed a wave of high-quality biosimilars. Indian and Chinese manufacturers now supply WHO-prequalified generics at <15% of originator prices-enabling national programs to treat 10x more patients per dollar.
- The HBV Functional Cure Race Is Accelerating: While HCV is largely “solved,” chronic HBV remains a therapeutic challenge. Over 30 candidates are now in clinical development-including siRNA (e.g., JNJ-3989), capsid assembly modulators (CAMs), and therapeutic vaccines-aiming for finite-duration therapy and HBsAg loss. This pipeline is re-energizing R&D investment and redefining long-term market potential.
“We’re transitioning from ‘treatment-as-lifelong-suppression’ to ‘treatment-as-cure’—but the real disruption is happening on the access front,” says Dr. Anjali Mehta, Principal Consultant, Infectious Disease Therapeutics Practice at Introspective Market Research.
“Biosimilars have democratized HCV cure in Southeast Asia and Africa. Now, the same model is being applied to tenofovir and entecavir for HBV. The next breakthrough won’t just be scientific—it’ll be logistical: point-of-care RNA testing, AI-driven patient triage, and drone-delivered antivirals in remote districts. That’s where the 4.9% CAGR in APAC is coming from-not just more drugs, but smarter delivery.”
Regional Deep Dive: North America Leads, but Asia Pacific Is the Growth Catalyst
North America (43.7% Market Share)
The U.S. remains the revenue anchor-supported by robust reimbursement (Medicare Part D, Medicaid expansion), premium pricing for novel regimens (e.g., Vosevi®, Vemlidy®), and integrated care models in managed Medicaid plans targeting high-risk populations (PWID, incarcerated individuals, migrants). Recent CMS coverage expansions for reflex HCV RNA testing post-antibody screen are expected to add 350,000+ treatable cases annually by 2027.
Asia Pacific (Projected CAGR: 4.9%)
APAC is the epicenter of both disease burden and market evolution:
- India: Launched National Viral Hepatitis Control Programme (NVHCP) with free HBV/HCV testing and treatment at 1,200+ centers; Zydus Cadila’s Sofosbuvir + Velpatasvir combo now priced at USD 180/course (vs. USD 26,000 originator launch).
- China: Scaling up HBV treatment under Healthy China 2030, with domestic NUCs capturing >85% volume share; Gilead’s Vemlidy approved for adolescents in 2025, expanding addressable population.
- Egypt & Pakistan: Mass HCV elimination campaigns using locally manufactured DAAs have treated >7 million patients since 2018-proving scalable cure is possible in resource-limited settings.
Africa & Middle East (High Unmet Need, Rising Momentum)
While fragmented, regions like Egypt (post-elimination maintenance), Morocco (new national HBV vaccination + treatment rollout), and South Africa (NHI formulary expansion) are emerging as strategic growth corridors-especially for affordable fixed-dose combinations.
Segmentation Analysis: Where the Value Flows
By Drug Class (2023 Share & Outlook)
| Segment | Key Agents | Clinical Role | Market Dynamics |
| Direct-Acting Antivirals (DAAs) | Sofosbuvir, Velpatasvir, Glecaprevir/Pibrentasvir | HCV cure (8–12 weeks) | Dominant segment; biosimilars driving volume surge; pan-genotypic regimens (>95% SVR) now standard |
| Nucleos(t)ide Analogues (NUCs) | Tenofovir (TAF/TDF), Entecavir, Lamivudine | First-line HBV suppression | High-volume, low-margin; TAF gaining share due to renal/bone safety; generics >70% of APAC volume |
| Interferons | Pegylated IFN-α | Legacy HBV/HCV therapy | Rapidly declining (<8% share) due to poor tolerability (flu-like symptoms, depression) |
| Immune Modulators | Thymosin-α1, novel checkpoint inhibitors (preclinical) | Adjunctive HBV functional cure | Niche today, but high strategic interest; potential to enable finite therapy |
By Disease Type
- Hepatitis B Drugs (52% share): Driven by chronicity (lifelong therapy), high prevalence in Asia/Africa, and premium NUCs (e.g., Vemlidy® at USD 12,000/year list price in U.S.).
- Hepatitis C Drugs (38%): Volume-driven by curative regimens; growth now tied to screening expansion and reinfection management in high-risk cohorts.
- Hepatitis A/D Drugs (10%): Smaller acute/niche markets-Hep A largely prevented by vaccine; Hep D (HDV) seeing renewed interest with bulevirtide (Hepcludex®) approval in EU (2024).
Innovation & Cost-Efficiency: How Leaders Are Expanding Access Without Sacrificing Margins
- Gilead Sciences (USA)-Q3 2025: Launched Vemlidy® Access Program in 12 LMICs-offering tenofovir alafenamide (TAF) at USD 48/year (vs. USD 1,200 in India private market)-paired with AI-powered adherence tracking via SMS.
- Dr. Reddy’s Laboratories (India)-Partnered with FIND and WHO to co-develop HepCheck™, a USD 2.50 dried-blood-spot (DBS) HBV DNA quantification kit-enabling decentralized viral load monitoring in rural clinics.
- Zydus Cadila (India)-Received WHO PQ for ZyHep-C® (sofosbuvir/velpatasvir), now supplying Egypt, Nigeria, and Bangladesh at USD 150/course-enabling treatment of 1 million+ additional patients in 2025 alone.
- AbbVie Inc.-Advanced ABBV-032 (siRNA + CAM combo) into Phase IIb for HBV functional cure-targeting HBsAg loss in >40% of patients after 48 weeks.
Cost-Optimization Strategies Gaining Traction
- Tender-Based Procurement: National programs (e.g., India, Egypt) bundling testing + treatment in single bids—reducing per-patient cost by 60–75%.
- Fixed-Dose Combinations (FDCs): Reducing pill burden and improving adherence (e.g., tenofovir/lamivudine for HBV; sofosbuvir/daclatasvir for HCV).
- Task-Shifting to Mid-Level Providers: Nurse-led HCV treatment in Thailand and Rwanda achieving >97% SVR—freeing hepatologists for complex cases.
- Digital Therapeutics Integration: App-based education and reminder systems cutting loss-to-follow-up by 33% in pilot programs (Brazil, Vietnam).
Cross-Stakeholder Benefits
- For Patients: Curative or suppressive therapy preventing cirrhosis, liver cancer, and transmission
- For Payers: Cost-offset via avoided transplants (1 liver transplant = 300+ DAA courses) and reduced hospitalizations
- For Governments: Progress toward WHO 2030 goals (90% diagnosed, 80% treated)—enhancing health security and productivity
- For Manufacturers: Volume growth in biosimilars; premium pricing for novel HBV functional cures; ESG credibility via access programs
Unlock Strategic Intelligence: Download the Full Report
The Hepatitis Drugs Market Report (2024–2032),by Introspective Market Research delivers 230+ pages of actionable insights for pharma strategists, public health planners, and investors:
- Country-level treatment coverage maps, pricing benchmarks (USD/patient/year), and reimbursement pathways
- Pipeline tracker: 32 HBV functional cure candidates (by mechanism: siRNA, CAM, TLR agonists, vaccines)
- Biosimilar landscape analysis: 18 WHO-prequalified DAAs/NUCs, with bioequivalence data and manufacturing capacity
- Access models deep-dive: Egypt’s elimination success, India’s NVHCP rollout, Rwanda’s nurse-led care
- Competitive positioning: Market share by region, payer mix, and molecule lifecycle status
Download a Free Sample Report:
https://introspectivemarketresearch.com/request/20191
About Introspective Market Research
Introspective Market Research(IMR) is a globally trusted provider of strategic intelligence across pharmaceuticals, vaccines, and global health. Our team of medical doctors, pharmacoeconomists, and health policy analysts combines primary interviews with 350+ clinicians, payers, and program managers-with real-world claims data, regulatory filings, and epidemiological modelling-to deliver insights that inform life-saving decisions.
We don’t just forecast markets—we help eliminate disease.
Media Contact
Dr. Neha Patel
Senior Director, Global Health Communications
Introspective Market Research
Call:- +91 91753-37569.
info@introspectivemarketresearch.com